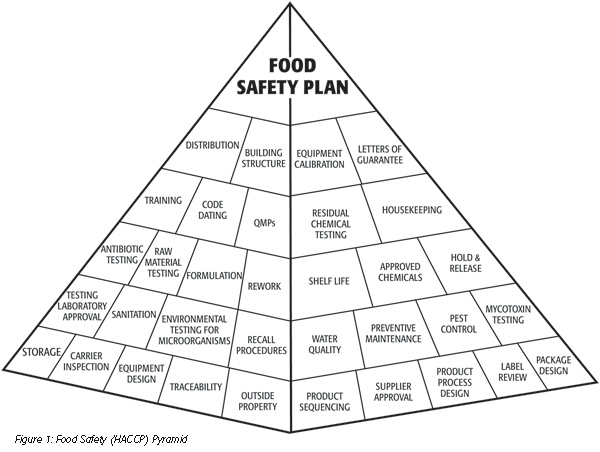
. Letters of guarantee. Container/truck inspection. Hold and release. Label review for instructions, (e.g. “Keep Refrigerated”). Pallet controls All raw materials should be purchased from an approved supplier and to up-to-date specifications. All raw materials should be kept separate from finished products. You may not need to write a guarantee letter from scratch. Many agencies provide a form, called a guarantor form. Sample of guarantee letter (letter head) consulate general of belgium 9/f., st.john’s building, 33, garden road, central, hong kong. Food Safety Letters & Audit Certificates. Food safety letters & audit certificates. When performing the HACCP 01 procedure and reviewing the records of this prerequisite program, the. Does the company have letters from suppliers indicating what they are doing to. Establishment requires suppliers to submit a letter of guarantee documenting the application of at least one intervention step (e.g., an antimicrobial treatment. HACCP Plan – Slaughter. Supplier may provide a letter of guarantee stating that the hazard should not be present. On the other hand, a history of outbreaks or contamination related to a hazard would mean that the hazard IS reasonably likely to occur. No Letter of guarantee from wax supplier ensuring that wax is food grade. Physical - None No SSOP makes hazard unlikely to occur. Scalding, Picking, Head Removal, Singeing Biological – Presence or growth of pathogens (see list above) Yes (Presence) Yes (Growth) Raw poultry is a known source of pathogens. Hazard will be controlled.
December 2007

Section 303 of the Federal Food, Drug, and Cosmetic Act
Haccp Letter Of Guarantee Templates Template
Section 303 of the Federal Food, Drug, and Cosmetic Act (the Act) provides penalties for persons who violate certain provisions of the Act. Section 303, paragraph (c) of the Act states that no person shall be subject to the penalties of subsection (a)(1) for having received, or proffered delivery of, adulterated or misbranded food additives if he has established a good faith guarantee from whom he received the articles. This paragraph was included in the 1958 amendments to the Federal Food, Drug and Cosmetic Act and remains the legal basis for the 'letter of guaranty' supplied by many manufacturers to their clients. Title 21 CFR 7.12 Guaranty and Title 21 CFR 7.13 Suggested Forms of Guaranty, provide example language for such a letter of guarantee.
